Certified lab testing: ensure reliable workplace screening
TL;DR:
- Only SAMHSA-certified labs provide legally defensible workplace drug test results.
- Certification involves rigorous performance testing, inspections, and ongoing compliance.
- Proper documentation and confirmatory testing are essential to ensure results withstand legal challenges.
Many employers assume that any positive drug test result is legally airtight. It isn’t. The type of lab that processes your sample determines whether that result will hold up in court, survive a federal audit, or protect your organization when an employee disputes a termination. Only testing conducted by a federally certified laboratory meets the forensic standards required for true workplace compliance. This guide breaks down what certified lab testing actually means, how the certification process works, what happens inside those labs, and how you can make the right call for your specific workplace situation.
Table of Contents
- What certified lab testing means (and why it matters)
- How certified labs earn and keep their status
- Inside the certified lab testing process: Key steps and safeguards
- What can go wrong: Limitations, disputes, and special cases
- Making the right choice: When certified lab testing is essential
- Perspective: Why process and paperwork matter as much as science
- Get started with certified lab testing you can trust
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Legal defensibility | Only certified lab tests hold up in court and regulatory hearings for workplace matters. |
| Rigorous certification | Labs must pass strict federal testing and inspection to gain and keep certified status. |
| Chain-of-custody procedures | Certified labs use exact sample tracking, confirmation methods, and MRO review for reliable results. |
| Minimized error | Confirmatory testing by certified labs cuts the risk of false positives and negatives dramatically. |
| Employer risk reduction | Choosing certified lab testing protects your organization from costly compliance mistakes. |
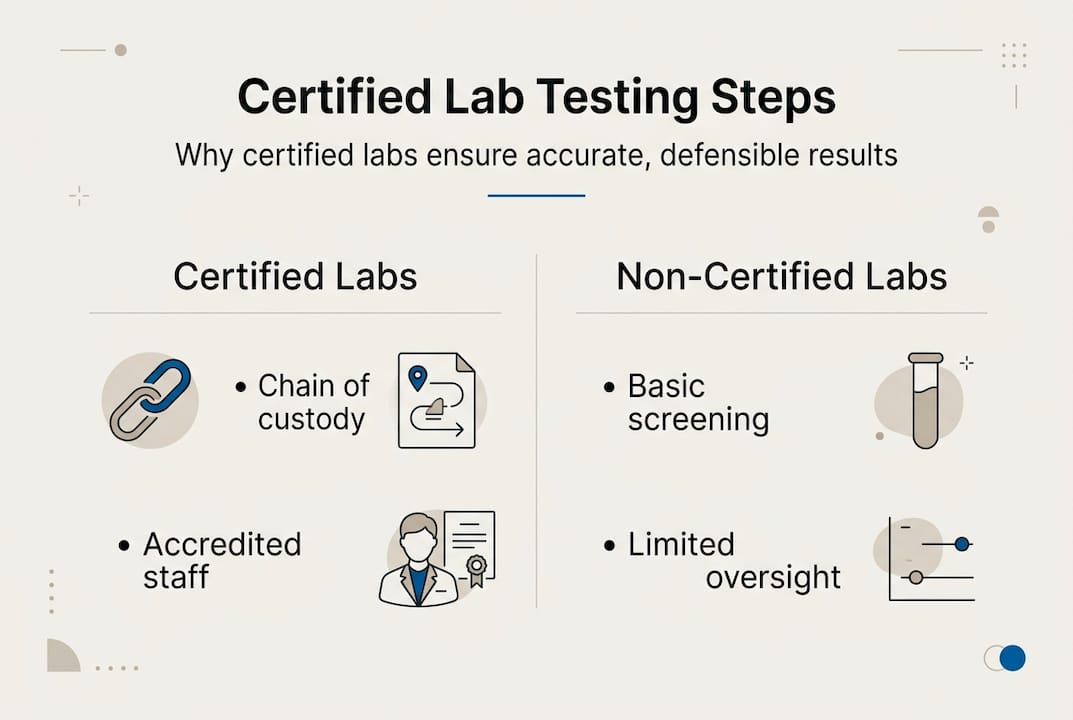
What certified lab testing means (and why it matters)
Not all labs are built the same, and the differences are not minor. Certified lab testing refers to drug and alcohol testing conducted by laboratories certified under SAMHSA’s NLCP for federal workplace drug testing programs. SAMHSA stands for the Substance Abuse and Mental Health Services Administration, and its National Laboratory Certification Program sets the gold standard for forensic workplace testing in the United States.
These standards exist because workplace drug testing is not just a health matter. It is a legal matter. When you use results from a SAMHSA-certified lab, you are producing forensic-grade evidence that can withstand scrutiny in arbitration, court proceedings, and federal audits. That is a fundamentally different product than a clinical diagnostic result.
Here is where many HR teams get tripped up. CLIA-certified labs, which handle most routine medical testing like blood panels and urinalysis for clinical purposes, are excellent at what they do. But clinical lab certification does not include the chain-of-custody documentation, confirmatory testing requirements, or Medical Review Officer involvement that forensic workplace testing demands. The difference between SAMHSA and CLIA labs is not a technicality. It is the line between a result you can act on and one you cannot defend.
Scenarios where certified lab testing is non-negotiable include:
- DOT-regulated positions such as commercial drivers, pilots, and railroad workers
- Federal contractor roles subject to the Drug-Free Workplace Act
- Safety-sensitive jobs in industries like construction, healthcare, and energy
- Court-ordered or probation-related testing where legal defensibility is required
- Employment terminations or disciplinary actions that could face legal challenge
“Only results from SAMHSA-certified labs are legally defensible for federally regulated workplace drug testing programs. Using a non-certified lab in these contexts exposes employers to serious compliance and legal risk.”
If your organization operates in any regulated industry, or if you ever anticipate that a test result might be disputed, certified lab testing is not optional. It is your protection.
How certified labs earn and keep their status
Earning SAMHSA certification is not a one-time paperwork exercise. The bar is genuinely high, and that is intentional. Certification requires three rounds of performance testing plus an on-site inspection before a lab can even begin processing workplace samples. After that, labs must pass quarterly performance testing and periodic on-site inspections to maintain their status.
The result? As of 2025, only about 18 labs nationwide hold current HHS certification. That number is not a sign of a niche market. It reflects how rigorous the requirements actually are.
Here is a summary of what the process looks like:
| Stage | Requirement | Frequency |
|---|---|---|
| Initial certification | 3 rounds of performance testing | One-time |
| Initial certification | On-site inspection | One-time |
| Ongoing maintenance | Quarterly performance testing | Every 3 months |
| Ongoing maintenance | Periodic on-site inspections | As scheduled by HHS |
| Ongoing maintenance | Proficiency testing panels | Continuous |
The numbered steps a lab must complete to reach certification status:
- Submit an application to the HHS/SAMHSA program
- Pass the first round of proficiency testing with blind samples
- Pass the second and third rounds under monitored conditions
- Undergo and pass a full on-site inspection by federal reviewers
- Maintain quarterly proficiency testing with no critical failures
- Accept periodic unannounced or scheduled re-inspections
This process filters out labs that cannot consistently deliver accurate, traceable results. When you send a sample to a certified lab, you are not just paying for chemistry. You are paying for a documented quality system that has been independently verified multiple times.
Pro Tip: Always verify a lab’s current certification status directly on the official HHS list before signing any testing contract. Certification can lapse, and an expired certification offers you no legal protection.
Inside the certified lab testing process: Key steps and safeguards
Understanding how drug tests work inside a certified lab explains why the results are so reliable. The process is not just a single chemical reaction. It is a multi-step, documented workflow designed to eliminate error and ensure traceability.
The standard workflow follows this path: sample collection with chain-of-custody paperwork, initial immunoassay screening, confirmatory GC/MS or LC-MS/MS for any presumptive positives, and final review by a certified Medical Review Officer, known as an MRO. Each step is documented, and every handoff of the sample is recorded.

| Feature | Certified (SAMHSA) lab | Non-certified (CLIA) lab |
|---|---|---|
| Chain of custody | Required and documented | Not required |
| Confirmatory testing | Mandatory for positives | Rarely performed |
| MRO review | Required | Not included |
| Legal defensibility | Yes | No |
| Adulteration testing | Required | Not standard |
The role of the Medical Review Officer is one of the most underappreciated safeguards in the process. An MRO is a licensed physician trained to interpret drug test results in context. They review every positive result, contact the donor to check for legitimate medical explanations, and make the final call before results reach the employer. This step protects both the employee from unfair outcomes and the employer from acting on an incorrect result.
Common sources of error that the certified process eliminates:
- Sample mix-ups prevented by strict chain-of-custody labeling
- Contaminated specimens caught by adulteration and validity testing
- False positives from immunoassay resolved by mandatory confirmation
- Undocumented handoffs eliminated by custody paperwork at every transfer
- Unreported prescription interference addressed through MRO review
Pro Tip: Never make an employment decision based solely on an initial immunoassay screen. That result is presumptive, not confirmed. Only act after the full certified lab process, including MRO sign-off, is complete.
What can go wrong: Limitations, disputes, and special cases
Even with the best processes in place, drug testing is not immune to complexity. Immunoassay screens, the first-line test used in certified labs, are fast and cost-effective but can generate false positives in certain cases. Common culprits include ibuprofen triggering a THC positive, poppy seed consumption flagging opioids, and certain antihistamines appearing as amphetamines.
This is exactly why confirmatory testing using GC-MS or LC-MS/MS technology is mandatory in certified labs for any presumptive positive. These methods are highly specific and accurate, identifying the exact compound rather than a class of substances. Confirmatory testing reduces false positive rates from a potential 5 to 10 percent with immunoassay alone to near zero.
“Immunoassay screens can produce false positives at rates of 5-10% in some populations. Confirmatory GC-MS or LC-MS/MS testing effectively eliminates this risk, making it the standard for any result used in employment decisions.”
Scenarios most prone to error or dispute:
- Poppy seed ingestion producing opioid positives on immunoassay
- Over-the-counter medications like ibuprofen or pseudoephedrine
- Passive cannabis exposure in states with legal recreational use
- Specimen adulteration attempts using household chemicals
- Diluted samples from excessive water intake before collection
Certified labs are equipped to handle all of these. Adulteration and dilution testing is built into the certified process, and split specimen procedures allow employees to request an independent retest of their original sample. This transparency is not just fair. It is a legal requirement in federally regulated programs.
Making the right choice: When certified lab testing is essential
Not every employer is subject to federal drug testing mandates, and that creates some flexibility. But flexibility is not the same as freedom from risk. Here is how to think through your decision.
Scenarios where certified lab testing is non-negotiable:
- Any role regulated by the DOT, FAA, FRA, or other federal agencies
- Positions covered by the Drug-Free Workplace Act
- Safety-sensitive jobs where impairment could cause serious harm
- Situations involving legal action, termination disputes, or arbitration
- Pre-employment screening for roles with federal contract requirements
A practical decision checklist for HR teams:
- Identify whether your industry is federally regulated
- Review your state’s drug testing laws, especially for cannabis
- Assess whether any roles are safety-sensitive by nature
- Determine if your drug-free workplace policy could face legal challenge
- Confirm that your current testing provider uses a SAMHSA-certified lab for all regulated positions
- Document your testing program in writing, including lab credentials
For private employers in non-regulated industries, CLIA labs may be acceptable for general wellness or internal policy purposes. But the moment a result could influence a termination, a workers’ compensation claim, or a legal proceeding, you need certified lab results.
Pro Tip: Cannabis legalization at the state level does not override federal testing requirements. If your employees are in DOT-regulated roles, federal drug testing laws still apply regardless of state law. Confirm your program meets both layers of compliance.
Perspective: Why process and paperwork matter as much as science
After working with employers across many industries, the pattern is clear: most compliance failures in drug testing are not caused by bad science. They are caused by skipped steps and missing documentation.
HR teams often choose a testing vendor based on price or turnaround time, without asking whether the lab is SAMHSA-certified. That decision seems harmless until an employee challenges their termination, and the employer discovers their test results carry no legal weight. The chemistry might have been accurate. But without the chain-of-custody paperwork, the MRO sign-off, and the certified lab credential, the result is indefensible.
Certified labs produce documentation as much as they produce results. That paperwork is what survives an audit, holds up in arbitration, and protects your HR policy when it is tested under pressure. Confirmatory testing for compliance is not a luxury. It is the foundation of a program that actually works when you need it most. The science matters. The process matters more.
Get started with certified lab testing you can trust
Understanding the stakes is the first step. Acting on that knowledge is what protects your organization. Countrywide Testing partners with a national network of SAMHSA-certified laboratories to give employers and HR teams access to reliable, legally defensible drug and alcohol screening.
Whether you need lab testing services for regulated roles, pre-employment screening, or ongoing workplace compliance programs, Countrywide Testing has certified options ready for your needs. For teams that also want on-site screening capability, our 12-panel drug test cup is a practical complement to lab-based confirmation. Reach out today and build a testing program that holds up when it counts.
Frequently asked questions
What does it mean if a lab is SAMHSA certified?
A SAMHSA-certified lab meets federal forensic standards for workplace drug testing, meaning results are legally defensible and suitable for regulated employment decisions.
Is there a difference between SAMHSA and CLIA certification?
Yes. SAMHSA certification covers forensic workplace testing with chain-of-custody and MRO requirements, while CLIA certification covers medical diagnostics without those legal safeguards.
How can I check if a lab is currently certified?
You can search the official HHS-certified lab list maintained by SAMHSA online. As of 2025, only approximately 18 labs hold current certification.
Why should employers use certified labs for drug testing?
SAMHSA-certified labs ensure test results meet federal standards for regulated and safety-sensitive roles, protecting employers from compliance failures and legal exposure.
What if an employee disputes their positive result?
Certified labs support split specimen retesting, and a Medical Review Officer reviews every positive result before it reaches the employer, ensuring disputes are handled fairly and transparently.
